Quality assurance has been around much longer than the CLIA regulations. It is an ongoing, comprehensive program which analyzes every aspect of an entire operation; it involves determining a quality goal, deciding whether or not the goal has been achieved, and implementing corrective action if the goal has not been reached.
In the laboratory, quality assurance involves the entire testing process: pre-analytical, analytical (testing), post-analytical processes, and staff performance. The CLIA regulations (Subpart K) addresses laboratory requirements and quality assurance systems. Medical decisions are made based on lab results performed in the office and at reference laboratories. Where the test is performed should not negate the significance of accurate and reliable results. Accurate test results are crucial to patient care.

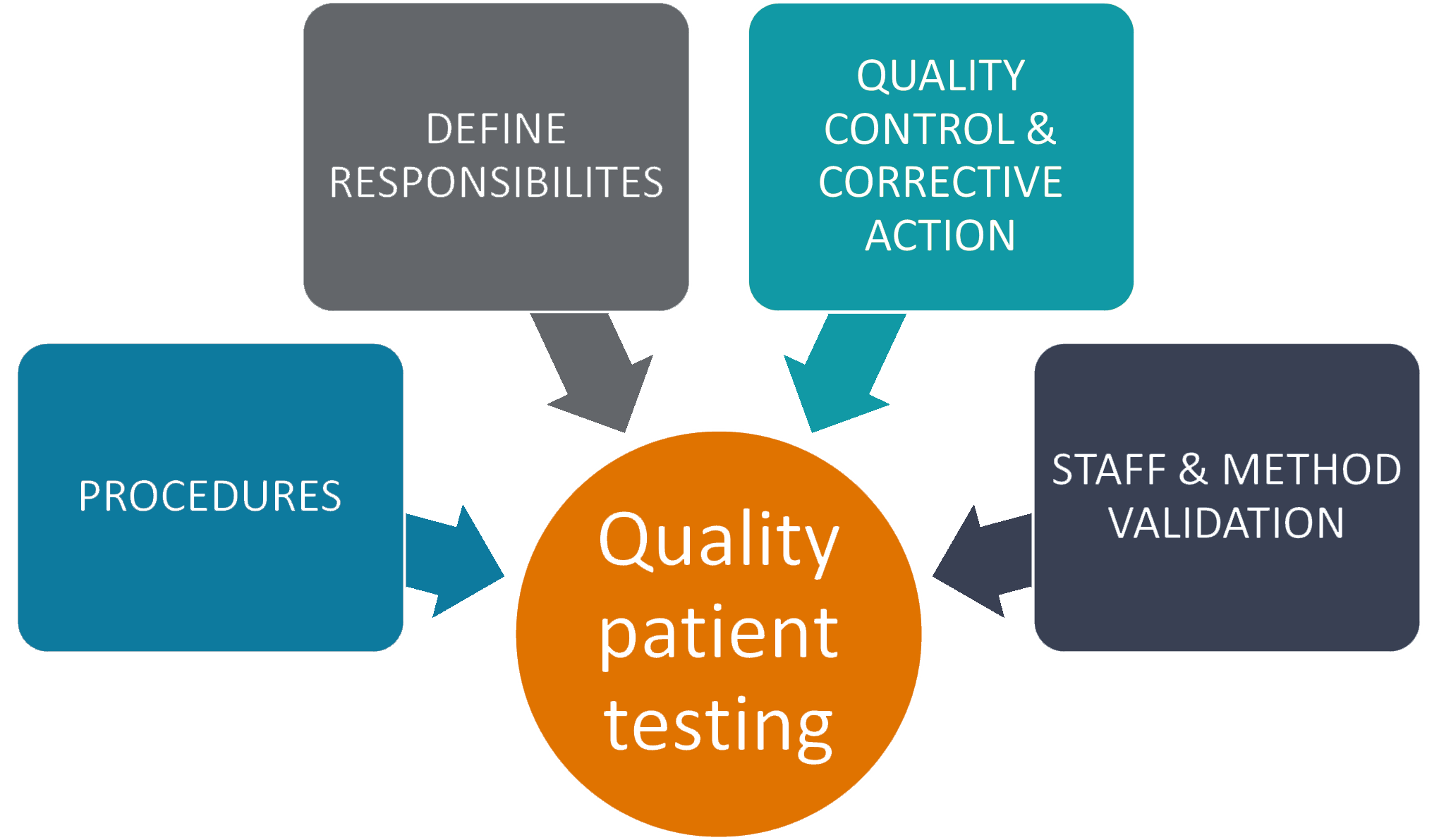
Assessing testing quality requires planned and systematic monitoring to test processes. Conducting these activities can lead to reduced errors, improved patient outcomes, improved patient and employee safety, and reduced costs. Both internal and/or external mechanisms for quality assurance may be used depending on the needs, resources, and practices of the testing site.
First: Establish laboratory procedures for each step of the testing processing, include components of testing such as, but not limited to:
Second: Define administrative responsibilities to laboratory management. Consider things such as:
Third: Specify, in writing, laboratory’s investigation plan and who will be responsible for follow through and implementation of correction action when problems or errors are identified. Document all quality assurance activities. As the saying goes, if it is not written down, it didn’t happen.
Fourth: High-quality patient testing requires verification of staff competency and test methods employed in your medical lab.
Staff competencies should assess their knowledge, skills and abilities needed to perform patient testing successfully. Staff assessments should be documented and performed annually after the first year of employment. During first year of employment, competency assessment is required semiannually.
Competency assessment must include the following 6 components for each test or test platform:
CLIA does not require competency assessment of staff for waived testing, however ensuring accurate results is essential to make the right patient diagnosis. Staff assessments are necessary to ensure staff are following manufacturers’ instructions. Keep in mind, any variation from manufacturer’s instructions of waived tests will negate waived status and test will be subject to all regulations as a moderate complex test.
Laboratories with provider-performed microscopy (PPM) certificates are required to meet competency assessment in all 6 components. All testing personnel in PPM laboratories are required to have annual competency.
For detailed information from CMS regarding staff competency, visit CMS brochure What Do I Need to Do to Assess Personnel Competency.
Method verification of test procedures is required to ensure, by objective evidence, the instrument or test method consistently provides accurate patient results Method verification must be completed prior to reporting your first patient test result. CMS has a published guide for Verification of Performance Specifications to help navigate this regulation.
When should method verification be performed?
Who is responsible for ensuring verification is completed and meets criteria?
What performance specifications are included in verification?
How is test verification most commonly accomplished?
These materials can be used in multiple ways to verify accuracy, precision, and reportable range. The number of specimens needed for each analyte and for each part of the verification process may vary. The Laboratory Director and Technical Consultant/Supervisor are responsible for determining the appropriate number of samples. As a rule of thumb, 20 specimens spanning the reportable range for quantitative analytes. Five positive and five negative specimens for qualitative analytes.